অনুসন্ধান করুন
অভিধানের ভাষা নির্বাচন করুন
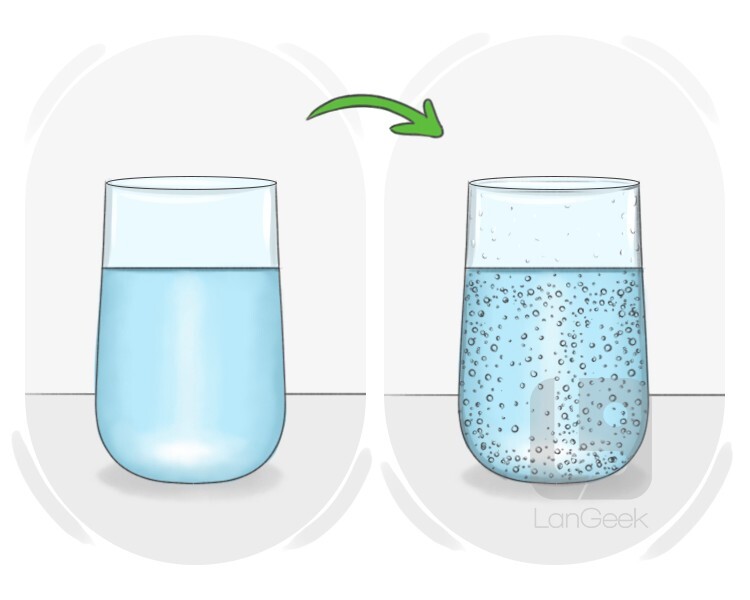
to carbonate
01
কার্বনেট করা, গ্যাস যোগ করা
to add carbon dioxide to something, often to make it fizzy or create a chemical change
Transitive: to carbonate a liquid
ব্যাকরণগত তথ্য
রূপগত গঠন
উৎপন্ন
কর্মসূচক ক্রিয়া
নিয়মিত
বর্তমান কাল
carbonate
৩য় পুরুষ একবচন
carbonates
বর্তমান কৃদন্ত
carbonating
সরল অতীত
carbonated
অতীত কৃদন্ত
carbonated
উদাহরণ
The brewing process has carbonated the beer naturally.
ব্রিউইং প্রক্রিয়াটি বিয়ারকে স্বাভাবিকভাবে কার্বনেটেড করেছে।
02
কার্বনেট করা, কার্বনেট যৌগে রূপান্তর করা
to convert a substance into a carbonate compound by reacting with carbon dioxide or a carbonate source
Transitive: to carbonate a substance or solution
উদাহরণ
By reacting sodium hydroxide with carbon dioxide, it is possible to carbonate sodium hydroxide.
সোডিয়াম হাইড্রোক্সাইডকে কার্বন ডাইঅক্সাইডের সাথে বিক্রিয়া করিয়ে, সোডিয়াম হাইড্রোক্সাইডকে কার্বনেট করা সম্ভব।
Carbonate
01
কার্বোনেট, কার্বনিক অ্যাসিডের লবণ বা এস্টার (CO3 অ্যানায়ন ধারণকারী)
a salt or ester of carbonic acid (containing the anion CO3)
ব্যাকরণগত তথ্য
প্রাণবাচকতার অবস্থা
জড়
রূপগত গঠন
যৌগিক
গণনীয়
বহুবচন রূপ
carbonates
শব্দতাত্ত্বিক গাছ
carbonated
carbonation
decarbonate
carbonate
carbon




























